|
12/7/2023 0 Comments Chlorine atomic massElemental chlorine at high concentration is extremely dangerous, and poisonous to most living organisms. As a common disinfectant, elemental chlorine and chlorine-generating compounds are used more directly in swimming pools to keep them sanitary. Chlorine is used in the manufacture of a wide range of consumer products, about two-thirds of them organic chemicals such as polyvinyl chloride (PVC), many intermediates for the production of plastics, and other end products which do not contain the element. The high oxidising potential of elemental chlorine led to the development of commercial bleaches and disinfectants, and a reagent for many processes in the chemical industry. These crustal deposits are nevertheless dwarfed by the huge reserves of chloride in seawater.Įlemental chlorine is commercially produced from brine by electrolysis, predominantly in the chlor-alkali process. It is the second-most abundant halogen (after fluorine) and twenty-first most abundant chemical element in Earth's crust. In 1809, chemists suggested that the gas might be a pure element, and this was confirmed by Sir Humphry Davy in 1810, who named it after the Ancient Greek χλωρός ( khlōrós, "pale green") because of its colour.īecause of its great reactivity, all chlorine in the Earth's crust is in the form of ionic chloride compounds, which includes table salt. Carl Wilhelm Scheele wrote a description of chlorine gas in 1774, supposing it to be an oxide of a new element. However, the nature of free chlorine gas as a separate substance was only recognised around 1630 by Jan Baptist van Helmont. It is an extremely reactive element and a strong oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Pauling scale, behind only oxygen and fluorine.Ĭhlorine played an important role in the experiments conducted by medieval alchemists, which commonly involved the heating of chloride salts like ammonium chloride ( sal ammoniac) and sodium chloride ( common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and hydrochloric acid (in the form of aqua regia). Chlorine is a yellow-green gas at room temperature. 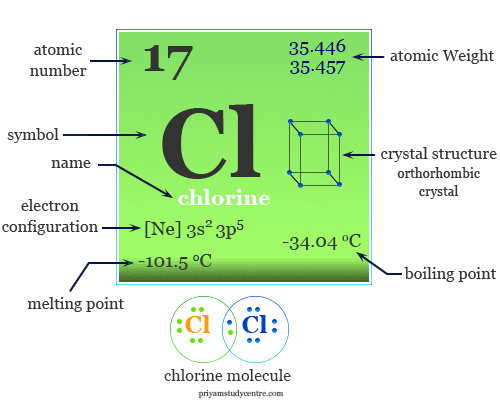
The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.Chlorine is a chemical element with the symbol Cl and atomic number 17. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. This site explains how to find molar mass. 
The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Ī common request on this site is to convert grams to moles. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. 
When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. These relative weights computed from the chemical equation are sometimes called equation weights.įinding molar mass starts with units of grams per mole (g/mol). In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
